LUMIC
Imagerie cellulaire, microscopie électronique, cytométrie en flux, cytométrie de masse, tri cellulaire et analyse multiplex pour la recherche fondamentale, clinique et translationnelle en biologie et sciences médicales.
LUMIC
Imagerie cellulaire, microscopie électronique, cytométrie en flux, cytométrie de masse, tri cellulaire et analyse multiplex pour la recherche fondamentale, clinique et translationnelle en biologie et sciences médicales.
LUMIC
LUMIC est le réseau des plateformes d’imagerie et de cytométrie de Sorbonne Université. Ses missions portent sur la formation, la recherche et le développement dans le domaine de l’imagerie cellulaire, de la microscopie électronique, de la cytométrie en flux et de la cytométrie de masse. LUMIC fournit un support technique et méthodologique pour la recherche fondamentale, translationnelle et clinique en biologie et sciences médicales. Ses atouts principaux sont de réunir des biologistes, médecins et physiciens, de promouvoir la réflexion au-delà des frontières et de favoriser le transfert d'outils développés à l'interface des disciplines vers la communauté scientifique et l'industrie.
LUMIC propose une offre de services intégrée et 1 800 m² de laboratoires accessibles aux utilisateurs sur site et externes, académiques et privés. La plateforme héberge le matériel permettant d'analyser des échantillons de la molécule à l'organisme entier, avec une résolution allant de l’angström au millimètre. Répartis sur 3 sites, ses physiciens développent des méthodologies, instruments et outils d’analyse de données sophistiqués pour répondre aux besoins spécifiques des utilisateurs. Les offres de services de la plateforme incluent la préparation d'échantillons, l'acquisition de données, leur traitement et analyse, ainsi que la formation dans quatre domaines clés : l’imagerie fonctionnelle, l’imagerie volumétrique, l’imagerie ultrastructurale, et la cytométrie en flux et de masse à haut débit.
Expertises et services
Imagerie fonctionnelle :
- Imagerie par fluorescence, de la molécule à l'organisme entier, imagerie de cellules, tissus et petits organismes vivants ou fixés,
- Imagerie de cellules vivantes et analyse de leur dynamique moléculaire (FRAP, FLIP, FDAP, photoactivation et photoconversion), imagerie calcique et biosenseurs,
- Microscopie confocale de super résolution,
- Imagerie macroscopique,
- Photomanipulation (photolésion et optogénétique),
- Imagerie spectrale et microspectroscopie confocale,
- Imagerie Raman confocale,
- Imagerie TIRF, PALM et STORM, suivi de particules uniques,
- Analyse d’interactions moléculaires par FRET et FLIM,
- Développement de flux de travail d'analyse de données.
Imagerie volumétrique :
- Clarification d'échantillons pour la microscopie confocale biphoton et à feuille de lumière,
- Imagerie confocale et à deux photons de tissus, organes et embryons,
- Microscopie à feuille de lumière,
- Visualisation, traitement et analyse de données avec Imaris et Arivis, déconvolution (confocal, biphoton, spinning disk, champ large), segmentation, analyse spatiale en 3D (colocalisation, suivi, ramifications), reconstruction 3D et lames virtuelles,
- Tomographie 3D en microscopie électronique : tomographie électronique et tomographie cryo-électronique en microscopie électronique à transmission (TEM), fraisage en série par FIB-SEM et array-tomography par microscopie électronique à balayage (SEM).
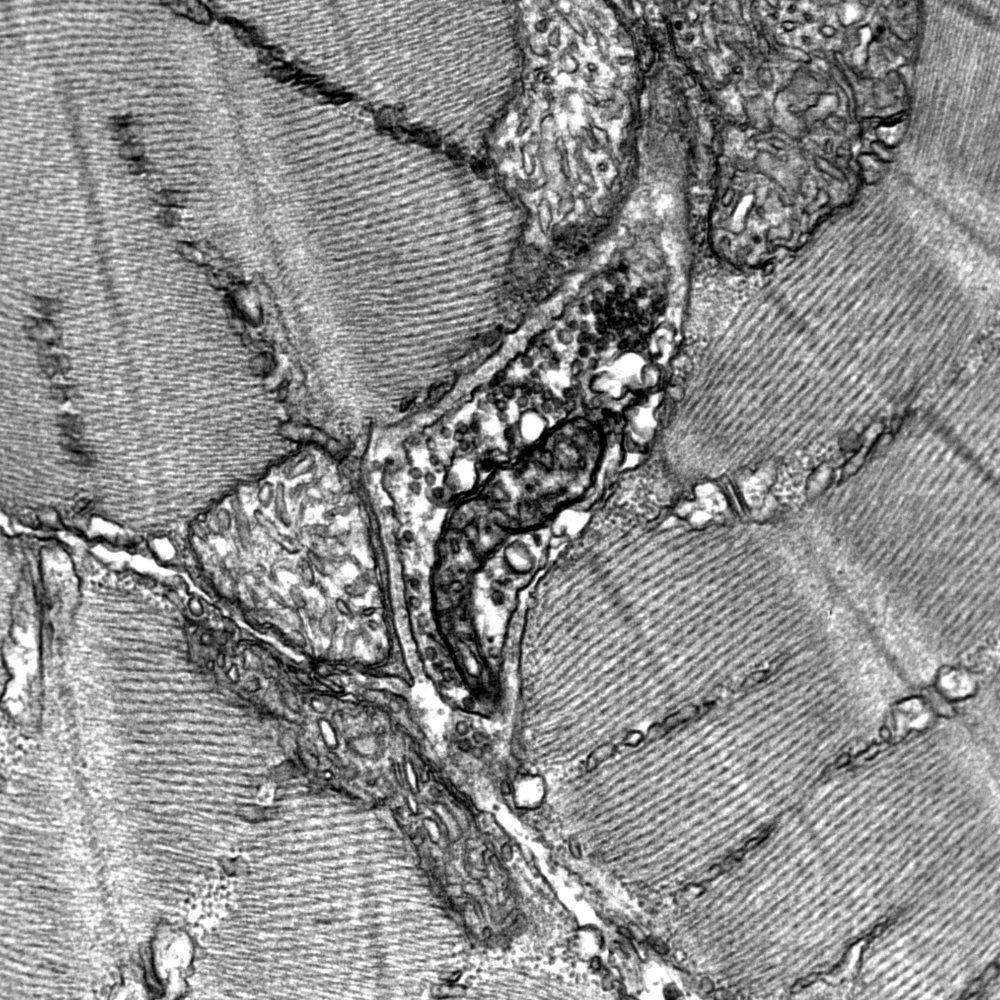
Imagerie ultrastructurale et analyse chimique par TEM :
- Préparation et observation d'échantillons conventionnels par TEM : ultrastructure, immunocytochimie et contraste négatif,
- Ultramicrotomie,
- Congélation à haute pression ou par plonge et substitution à froid,
- Cryo-microscopie,
- Cartographie chimique et spectroscopique par TEM ou STEM (STEM-HAADF),
- Spectroscopie aux rayons X à haute énergie de dispersion (XEDS),
- Spectroscopie et imagerie par perte d’électron de haute énergie (EELS/EFTEM).
Imagerie ultrastructurale et analyse chimique par SEM :
- Préparation d’échantillons et observation à haute résolution par SEM et cryo-SEM,
- Imagerie conventionnelle (électrons secondaires et rétro-dispersés),
- Spectroscopie aux rayons X à haute énergie de dispersion (XEDS),
- Préparation d’échantillons : séchage au point critique, fraisage par faisceau d'ions, fracturation par congélation et gravure par congélation, métallisation secondaire.
Imagerie ultrastructurale et analyse chimique par FIB-SEM :
- Préparation d’échantillon pour TEM et microscopie aux rayons X à transmission et balayage (STXM), fraisage en série,
- Tomographie 3D, Atlas 5.
Techniques à haut débit :
- Cytométrie en flux multiparamétrique (15 à 18 marqueurs) et acquisition de données,
- Analyse de données de cytométrie en flux multiparamétrique (logiciels et expertise),
- Analyse de la mort cellulaire, du cycle cellulaire et flux de calcium par cytométrie en flux,
- Tri cellulaire avec niveau de sécurité P2 disponible sur 2 sites, enrichissement par tri automatique de cellules magnétiques et tri FACS, microparticules, événements rares et dispositifs de clonage simultané pour 6 populations maximum,
- Analyse par cytométrie de masse (CyTOF) jusqu'à 30 marqueurs, traitement des données et analyse (SPADE),
- Multiplexage élevé : détection et quantification de protéines sécrétées ou provenant de lysat de cellules ou tissus,
- Développement de kits de détection multiplex,
- Imagerie spectrale de biomarqueurs multiplexés en champ clair ou fluorescence,
- Multiplexage virtuel : enregistrement d'images de sections sérielles immunoétiquetées, y compris de lames virtuelles,
- Imagerie à haut débit en champ clair ou fluorescence,
- Analyse d’images, lames virtuelles et tissue microarray (TMA),
- Analyse de cellules en temps réel (xCELLigence) pour étudier l'adhésion, la prolifération, l'invasion et la migration de cellules par impédancemétrie.
Moyens et équipements
Microscopes confocaux :
- Microscope inversé Leica TCS SP5 AOBS avec détecteur hybride et scanner résonant (IBPS),
- Microscope droit Leica TCS SP5 AOBS avec détecteur hybride (IBPS),
- Microscope droit Olympus FV1000 (ICM),
- Microscope inversé Zeiss LSM710 (CRC),
- Microscopes droit et inversé Olympus FV1000 (IDV),
- Microscope droit Leica TCS SP5 AOBS avec détecteur hybride (IFM),
- Microscope inversé Olympus FVi10x (IFM),
- Microscope droit Leica SPE(IBPS),
- Microscope inversé Olympus FV1200 (IDV),
- Microscope inversé Leica TCS SP8 AOBS avec détecteur hybride (ICM),
- Microscope droit Olympus FV1200 avec détecteur GaAsP (ICM).
Microscopes multiphotons :
- Microscope droit Zeiss 710 NLO avec détecteur GaAsP (ICM),
- Microscope droit Leica TCS SP8 MP5 avec détecteur hybride et scanner résonant (IBPS),
- Microscope droit Leica TCS MP5 avec détecteur hybride et scanner résonant (IFM),
- Microscope droit 3I avec scanner résonant, holographie et détecteurs GaAsP (ICM).
Microscopes spinning disk :
- Spinning disk droit Roper/Leica (IDV),
- Spinning disk inversé Roper MetaMorph (IBPS),
- Spinning disk inversé Leica (IFM),
- Spinning disk inversé Roper/Leica (UMS-StA),
- Spinning disk droit Roper/Leica (IDV),
- Spinnnig disk droit avec FRAP-photoactivation (IBPS),
- Microscope inversé Olympus IX83 et Andor DSD2 (Tenon),
- Spinning disk inversé Leica Yokogawa X Andor (ICM),
- Spinning disk droit Yokogawa W (ICM).
Microscopes à feuille de lumière :
- Ultramicroscope avec laser dans le rouge LaVision Biotec (IDV),
- Ultramicroscope LaVision Biotec (ICM),
- Microscope PhaseView (IBPS).
Scanners de lames :
- Scanner à fluorescence et lumière blanche Hamamatsu (IDV),
- Zeiss Axioscan Z1 (CRC).
Prototypes de microscopes :
- 2 microscopes à feuille de lumière pour l'imagerie du poisson zèbre (LJP),
- Microscope Raman confocal et Raman à pinces optiques (LJP),
- 3 microscopes optiques couplés à la microfluidique (LJP)
Equipements d’histologie :
- Automate immunhistochimie Dako (UMS-StA),
- Tissue microarrayer (UMS-StA),
- Système de marquage de lames automatisé IHC/ISH Roche Ventana Discovery XT (CRC).
Microscopes à haut débit :
- Microscope multispectral automatisé PerkinElmer Vectra (CRC),
- Microscope multispectral automatisé PerkinElmer Mantra (CRC),
- Microscope haut débit Beckton Dickinson BD855 (CRC).
Microscopes à dissection laser :
- Microscope à dissection laser AS-LMD Leica (CRC),
- Microscope à dissection laser PALM Zeiss (UMS-StA),
- Microscope à dissection laser Nikon Arcturus (IBPS),
- Microscope à dissection laser Leica DM6500B (IDV).
Microscopes électroniques et matériels pour la préparation d’échantillons :
- TEM Philips CM100 avec caméra CCD Gatan Orius (IFM),
- TEM Jeol 1010, 100 kV, avec caméra MegaView III (Tenon),
- TEM Jeol JEM 2100F, 200 kV, Gatan GIF et caméra CCD US4000, STEM-HAADF et BF, détecteur XEDS Jeol, cryo-TEM et tomographie (IMPMC),
- SEM Zeiss Ultra 55 avec système XEDS Bruker Quantax (IMPMC),
- FIB Zeiss Neon 40EsB avec système XEDS Bruker Quantax et tomographie Atlas 3D (IMPMC),
- TEM Jeol JEM 2100, 200 kV, caméra CCD Gatan US1000, STEM‑BF, détecteur XEDS Jeol, extension cryo-TEM et tomographie champ clair (IMPMC),
- TEM Jeol JEM 2100, 200 kV avec caméra Gatan Tridiem, tomographie et cryo-TEM (IBPS),
- TEM Hitachi 120 kV (ICM),
- FESEM à haute résolution Zeiss GeminiSEM 500 avec détecteur STEM, platine cryo Leica, microsystèmes et module de tomographie Zeiss Atlas 5 array tomography (IBPS),
- Appareil de fracturation par congélation et machines de revêtement Baltec BAF400T et ACE600 avec Leica VCT100 (IBPS),
- Système de congélation à haute pression Leica HPM100 (IBPS).
Analyseurs multilasers et trieurs de cellules :
- Cytomètre en flux Becton Dickinson LSR II (CRC),
- Cytomètre en flux Becton Dickinson LSR II (UMS-StA),
- Cytomètre en flux Beckman Coulter FC500 (IDV),
- Analyseur-trieur à 5 lasers L2 (UMS-PS),
- Cytomètre en flux Beckman Coulter Gallios (UMS-StA),
- Trieur Beckman Coulter MoFlo-Astrios standard (UMS-StA),
- Analyseur à 5 lasers (UMS-PS),
- Trieurs Becton Dickinson Aria II et Influx (CRC),
- Cytomètre en flux Miltenyi MACSquant Analyzer 10 (ICAN),
- Trieur Beckman Coulter MoFlo Astrios eQ (ICAN),
- Trieur magnétique Miltenyi AutoMacs (CRC).
Cytomètre de masse :
- CyTOF II mis à jour sur Hélios (UMS-PS).
Machines multiplex :
- Plateforme à haut multiplexage Luminex FLEXMAP 3D (CRC),
- Système multiplex Luminex MAGPIX (ICAN, IDV).
Matériel d’analyse d’interactions moléculaires :
- GE Healthcare Biacore 3000 (IBPS).



Comment soumettre un projet ?
Le site web de LUMIC présente un inventaire complet des techniques et du savoir-faire proposés par chacun des sites de la plateforme. Les équipements sont mis à disposition et réservables en ligne. Pour un service non collaboratif, vous devez envoyer votre demande à l’adresse lumic@services.cnrs.fr. Vous serez alors dirigé vers la méthodologie et les équipements appropriés, qui seront réservables en ligne dans un délai d'une à deux semaines. Pour les projets collaboratifs, vous devez adresser votre demande au comité de pilotage local du site concerné, qui définit les priorités des projets à sélectionner. Les délais de réalisation sont variables.
Les différents sites de la plateforme sont hébergés par divers organismes, universités, hôpitaux et fondations. Chacun de ces sites a établi une procédure interne pour recevoir ses utilisateurs, mais tous ont signé une charte des plateformes commune à Sorbonne Université. La détermination des coûts, la validation des tarifs et la facturation sont toujours effectuées par Sorbonne Université.
Exemple d'utilisation
Imagerie de super résolution confocale sur des échantillons complexes
Un projet initié par l’équipe de Katja Wassmann au Laboratoire de biologie du développement (LBD) sur le site IBPS visait à acquérir une image à très haute résolution du fuseau méiotique d'ovocytes de souris pour obtenir des informations qualitatives sur l'interface cytosquelette / kinétochore. Les techniques de super résolution telles que la 3D-SEM ne pouvaient être appliquées en raison de la complexité de l'échantillon.
Le site IPBS a développé une procédure incluant l’optimisation de la préparation des échantillons, de l'acquisition d'images et du traitement du signal, afin de surmonter les aberrations sphériques dues à l'opacité, l'épaisseur et la densité de marquage de l'échantillon. Le site IPBS a ainsi mis au point un processus permettant d’obtenir des images confocales de super résolution avec des résolutions comparables au 3D-SIM. La méthodologie développée est maintenant disponible pour tous les utilisateurs de LUMIC et extérieurs. Elle s’applique désormais à d’autres échantillons complexes, tels que les petits embryons.
Pour en savoir plus : Vallot A. et al. (2018). Tension-induced error correction and not kinetochore attachment status activates the SAC in an Aurora-B/C-dependent manner in oocytes. Current Biology, 28(1):130-139.
Contact
LUMIC
4 place Jussieu
75005 Paris
Région : Île-de-France + 33 (0)1 44 27 20 06
+ 33 (0)1 44 27 20 06
lumic@services.cnrs.fr
Site de la plateforme
THÉMATIQUES : Imagerie cellulaire, Autres
RESPONSABLES SCIENTIFIQUES :
Volker Bormuth
RESPONSABLES TECHNIQUES :
Olivier Renaud
TUTELLES : CNRS, Inserm, Sorbonne Université
LABELLISATION IBiSA : 2018
MOTS CLÉS : Imagerie fonctionnelle, STORM, PALM, TIRF, Clarification, Imagerie multiphotons, Déconvolution, Imagerie du vivant, Imagerie confocale, Imagerie spectrale, Raman, Microdissection laser, Imagerie calcique, Imagerie à feuille de lumière, Cytométrie multiparamétrique, Tri cellulaire, Cytométrie de masse, Microscopie électronique, TEM, SEM, Tomographie 3D, Cryotechniques, FIB-SEM, Cartographie chimique, STEM-HAADF
Fiche mise à jour en 2024










